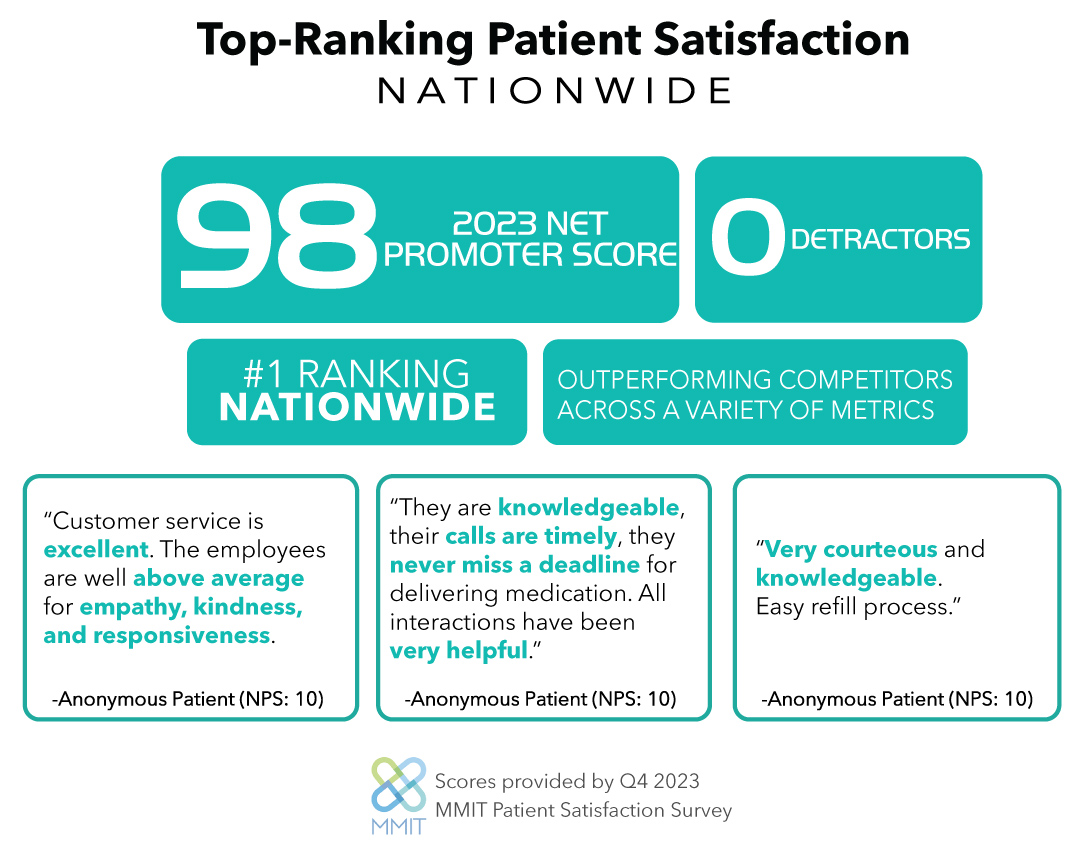
In their recently published white paper, RareMed’s Dr. Rami Chammas, Director of Pharmacy Operations, and Gannon Vanscoy, Vice President of Markets and Analytics, explore why transitioning an underperforming HUB or Non-Commercial Pharmacy program for a drug developed to treat a rare disease can be easy and in the best interest of patients, if certain considerations are taken into account.
Synopsis:
The term “white glove” has become one of the most overused words in the pharmacy services industry. Many HUB and non-commercial pharmacy (NCP) service providers claim white glove service as a key differentiator. However, the definition of white glove is variable, and different drugs demand different levels of HUB and NCP services.
White glove service promises often deteriorate when HUB or NCP services for rare diseases (populations less than 200,000 patients) are managed by partners that do not cater to the nuances of small populations. Programs can be too small to command attention in a world of million-patient populations, or too nuanced to fit with the standardized procedures that are acceptable for lower-touch medications. The inherent challenges of rare diseases demand a partner focused entirely on rare disease. A new breed of rare-focused HUBs has been developed to fill this marketplace gap, led by niche pioneer RareMed Solutions.
Proper planning is essential to addressing network gaps and pain points while managing a disruption-free transition. Benefits of a transition may include:
– Fewer prescription transfers, accelerating time to therapy
– Improved data visibility across the entire network
– Elimination of duplicate patients
– Streamlined processes
– Faster turnaround and dispensing
– Full separation of commercial and free drug dispensing
– New insights leading to the development of program enhancements